Surprise Move: AstraZeneca Agrees to Settle Proton Pump Inhibitor Lawsuits
In a surprise move, AstraZeneca PLC has agreed to a $425 million settlement to resolve product liability lawsuits in the US. A bellwether trial was scheduled for October which would have been followed by bellwether trials in November 2023 and January 2024. But AstraZeneca Plc has decided to resolve these cases immediately even ahead of the three remaining bellwether trials. This is an unusual move for a large company.
Most large organizations appear content during an MDL or class action lawsuit to drag things out for as long as they possibly can.
Only a single PPI case remains pending in a district court of Louisiana, for which a trial is scheduled for April 15, 2024.
The AstraZeneca Nexium Prilosec proton pump inhibitor drugs settlement was related to heartburn and stomach acid treatments, Nexium, and Prilosec, which some users claimed caused kidney damage and other disagreeable side effects.
This case has been ongoing for quite some time now. Since approximately 2016, there has been a significant increase in the number of lawsuits filed against AstraZeneca and other manufacturers of PPIs due to kidney damage. More than 15,000 plaintiffs have taken legal action against the manufacturers of Nexium, Prilosec, Prevacid, Protonix, and Dexilant, specifically regarding PPI-related kidney damage.
Plaintiff attorneys and the clients they represent must be looking forward to receiving just compensation for their work and for their suffering (respectively). And although this case has taken a few years to wind its way through the courts, relative to some MDL’s that have taken 18 years (like the Interchange fee lawsuits against Visa and Mastercard), 7 years to reach the settlement claims administration process doesn’t seem like such long time.
About the AstraZeneca Nexium Prilosec Proton Pump Inhibitor Drug Settlement
The AstraZeneca Nexium Prilosec proton pump inhibitor (PPI) drugs settlement has been a highly discussed and controversial topic in the pharmaceutical industry. This settlement involves one of the largest drug companies, AstraZeneca, and their popular PPI medications Nexium and Prilosec.

These drugs are commonly prescribed for conditions such as acid reflux, ulcers, and heartburn. However, despite their widespread use and popularity, these medications have faced numerous lawsuits due to serious side effects and alleged deceptive marketing practices by AstraZeneca.
AstraZeneca markets and sells pharmaceutical products in the United States, including a drug sold under the trade name Nexium. This settlement not only included monetary compensation for those affected but also required AstraZeneca to update their warning labels and provide more transparency in their marketing practices.
This settlement has major implications for both consumers and the pharmaceutical industry. It serves as a reminder that drug companies have a responsibility to properly inform patients about potential risks associated with their medications. It also highlights the importance of taking legal action when these responsibilities are not met.
In addition, this settlement may pave the way for similar settlements with other PPI drug manufacturers who have faced similar lawsuits. It also raises questions about the overall safety and effectiveness of PPI drugs and may lead to further research on alternative treatments for acid reflux and related conditions.
Overall, the AstraZeneca Nexium Prilosec PPI drugs settlement is a significant event in the pharmaceutical industry that has sparked important conversations about patient safety and corporate responsibility.
While it may not reverse the damage done to those affected by these medications, it serves as a reminder for companies to prioritize transparency and accountability in their practices.
Let’s explore what exactly happened with the AstraZeneca Nexium Prilosec proton pump inhibitor (PPI) drugs settlement and what it means for those affected.
When did the AstraZeneca Nexium Prilosec PPI Drugs Settlement Begin?
The AstraZeneca Nexium Prilosec PPI drugs settlement dates back to 2013 when the company was first accused of promoting their medications without proper warning labels about potential side effects.
These side effects included an increased risk of kidney disease, bone fractures, and even stomach cancer. As a result, thousands of lawsuits were filed against AstraZeneca by patients who claimed to have suffered from these adverse effects after taking their medication. People sued the makers of Nexium and Prilosec due to various health issues and injuries allegedly caused by these medications.
Some of the reasons for the lawsuits include:
- Kidney Injuries: Claimants in the lawsuits allege that the use of Nexium and Prilosec, both proton pump inhibitors (PPIs), can cause kidney damage and other renal problems.
- Deceptive Marketing: A national class action lawsuit was settled in 2015, accusing AstraZeneca, the manufacturer, of deceptive marketing practices related to Nexium. It was alleged that the company misled consumers into purchasing the more expensive Nexium instead of cheaper alternatives.
- Failure to Warn: Lawsuits also claim that the drug manufacturers knew as early as 2004 that Prilosec, Nexium, and other PPIs could cause kidney injuries but failed to adequately warn consumers or healthcare professionals about these risks.
- Personal Injury Claims: Numerous personal injury claims have been filed against the makers of Nexium and Prilosec, with plaintiffs alleging harm and seeking compensation for their medical expenses, pain and suffering, and other damages.
What is Nexium and Prilosec?
Nexium and Prilosec are from the proton pump inhibitor (PPI) class of drugs, which limit the production of acid in the stomach. They are commonly prescribed for acid reflux, gastroesophageal reflux disease (GERD), and other related conditions. These drugs work by blocking the enzyme in the stomach that produces acid, providing relief from symptoms such as heartburn, chest pain, and difficulty swallowing.
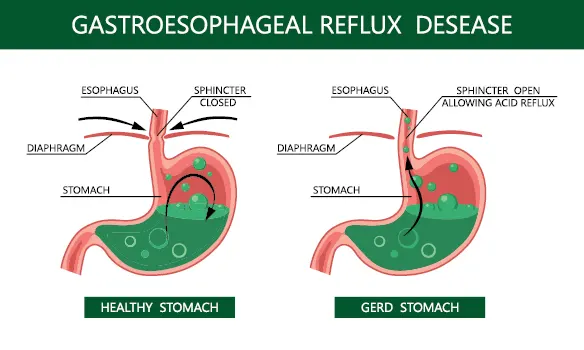
Nexium is AstraZeneca’s top-selling drug, earning over $3 billion a year in sales. Prilosec was also a top performer before it became available as an over-the-counter medication. Together, these medications have been used by millions of people worldwide.
However, despite their popularity and widespread use, Nexium and Prilosec have faced numerous lawsuits due to severe side effects that were not properly disclosed or warned about by the manufacturer.
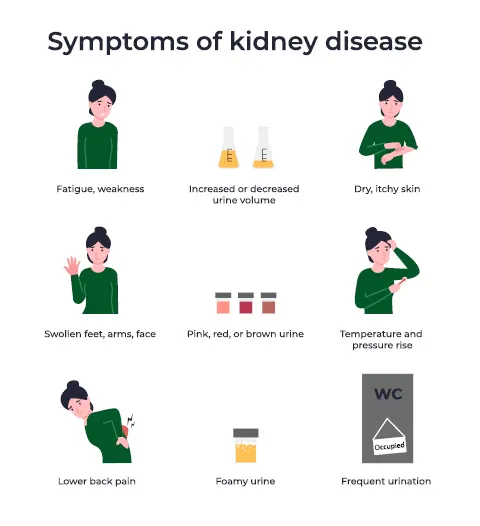
Nexium and Prilosec Linked to Chronic Kidney Damage
Since the initial FDA approval and introduction of PPIs like Prilosec and later Nexium in the 1990s, numerous scientific studies have established a correlation between prolonged PPI usage and significant negative health implications. These include but are not limited to bone fractures and pneumonia.
However, in 2015, new evidence surfaced that definitively established a direct correlation between long-term usage of PPIs and a substantial rise in the likelihood of enduring kidney injuries.
Recent studies have revealed that the prolonged usage of PPIs is linked to an independent 50% higher risk of developing chronic kidney disease.
Remarkably, even short-term use of PPIs has been shown to elevate the risk of CKD by 10%, according to one of these studies.
As this lawsuit has gone on, larger and more extensive studies have been conducted that have further proven that PPI’s cause Kidney Damage. This undoubtedly contributed to the company’s decision to settle these cases now rather than go through with the bellwether trials that are coming.
Chronic kidney disease is a progressive condition characterized by a gradual decline in kidney function. If left untreated, it can advance to end-stage kidney failure, a life-threatening condition requiring artificial filtration, dialysis, or a kidney transplant. CKD is strongly associated with a significantly elevated risk of death and cardiovascular events.
The Background of the Lawsuits
The lawsuits against AstraZeneca regarding their PPI medications allege that the company engaged in deceptive marketing practices by downplaying or omitting potential risks associated with taking Nexium and Prilosec.
These allegations suggest that patients were not properly informed about serious side effects such as kidney disease, bone fractures, and stomach cancer.
In addition to these accusations, there have also been claims of off-label promotion, meaning that AstraZeneca marketed these drugs for uses that were not approved by the FDA. This is a violation of the Federal Food, Drug, and Cosmetic Act.
As a result of these lawsuits, AstraZeneca faced numerous investigations and legal battles, eventually leading to the $7.9 billion settlement in 2020.
This is one of the largest pharmaceutical settlements in history, highlighting the severity and impact of this case.
This Is Not the First Time Makers of Proton Pump Inhibitors Have Faced Litigation
GSK, Pfizer, and Procter & Gamble have paid a combined $108 million to settle claims against their proton pump inhibitor treatments. These settlements have raised concerns about the overall safety and effectiveness of these drugs, prompting further research and investigations.
In addition to the AstraZeneca lawsuits, there have been numerous other lawsuits against makers of proton pump inhibitors. For example, in 2016, the FDA issued warnings about increased risks of kidney injury and bone fractures associated with PPI drugs.
This led to a wave of lawsuits against manufacturers such as Takeda Pharmaceuticals (makers of Dexilant) and Sanofi-Aventis (makers of Protonix). As a result, it is likely that we will see more settlements and legal actions against PPI drug makers in the future.
After years of litigation and ongoing investigations, AstraZeneca agreed to settle with the government for a whopping $7.9 billion in 2020. AstraZeneca LP agreed to pay the government $7.9 million to settle allegations that it engaged in a kickback scheme in violation of the False Claims Act.
As you can see, these manufacturers have been in hot water before for various allegations of misdeeds.
What Does This Mean for Consumers?
For those affected by Nexium or Prilosec, this settlement offers some form of justice through monetary compensation. However, it cannot reverse any damage or harm caused by taking these medications.
It also serves as a reminder for patients to be aware of potential side effects associated with any medication they are prescribed.
The Implications of the Settlement

The AstraZeneca Nexium Prilosec PPI drugs settlement has significant implications for both consumers and the pharmaceutical industry as a whole. For consumers, this settlement means that those who have suffered from serious side effects after taking Nexium or Prilosec may be eligible for compensation through the settlement.
Furthermore, this settlement serves as a reminder for pharmaceutical companies to prioritize transparency and accountability in their marketing and labeling practices. It also raises questions about the safety and effectiveness of PPI drugs in general, leading to further research and potential changes in prescribing practices.
For the pharmaceutical industry, this settlement may pave the way for similar settlements with other PPI drug manufacturers who have faced similar lawsuits. It also puts pressure on companies to ensure that their medications are properly tested and labeled before being brought to market.
Conclusion
The AstraZeneca Nexium Prilosec PPI drugs settlement has been a pivotal moment in the pharmaceutical industry, shedding light on the importance of transparency and accountability. While it cannot undo the harm caused to those affected by these drugs, it serves as a reminder for companies to prioritize patient safety above all else.
Post Settlement Funding for Attorneys Involved in the Nexium and Prilosec MDL
Post settlement funding can provide financial support to plaintiff attorneys involved in the AstraZeneca Nexium Prilosec proton pump inhibitor (PPI) drugs settlement. This form of funding allows attorneys to access a portion of their anticipated fees before the final settlement is paid out, providing them with much-needed resources for ongoing litigation expenses and other overhead costs.
With post settlement funding, attorneys can avoid cash flow issues and focus on advocating for their clients while awaiting the resolution of the case. As this is a large and complex settlement involving multiple parties, attorneys on the Prilosec and Nexium case may be forced to wait for years to receive their final fees from the AstraZeneca Settlement. Post settlement funding is a great way for plaintiff attorneys to get an advance on some of their fees now to continue funding their ongoing cases, or land new clients.

The length of time involved in the AstraZeneca Nexium Prilosec PPI drugs settlement case has likely had a significant impact on the business cash flow of plaintiff attorneys. With ongoing litigation expenses and overhead costs, it can be challenging for attorneys to maintain a steady cash flow during such a lengthy legal process.
This is where post settlement funding can be especially beneficial. By providing access to a portion of their anticipated fees before the final settlement is paid out, post settlement funding can help alleviate financial strain and allow attorneys to focus on advocating for their clients without worrying about day-to-day expenses.
Post Settlement Funding for Attorneys Involved in the AstraZeneca Nexium Prilosec PPI Drugs Settlement
If you are an attorney who has been involved in the AstraZeneca Nexium Prilosec proton pump inhibitor (PPI) drugs settlement, then you are likely aware of the challenges that come with representing clients in mass tort litigation.
The long wait for a settlement and the financial strain it can put on your law firm can be overwhelming. However, there is a solution that can help alleviate these burdens – post settlement funding.
What is Post Settlement Funding?
Post settlement funding, also known as litigation funding or legal funding, is a financial tool designed specifically for attorneys who represent clients on a contingency fee basis. Post settlement funding provides immediate funding to law firms with cases that have reached a settlement agreement but will have delays until the contingency fees are distributed.
This type of funding is not a traditional loan, as it does not require monthly payments or any ongoing payments.

How Can Post Settlement Funding Benefit Attorneys in the AstraZeneca Nexium Prilosec PPI Drugs Settlement?
Post settlement funding can provide attorneys with financial stability and flexibility during the long wait for the defendant to pay what they owe.
Here are some benefits that post settlement funding offers specifically for attorneys involved in the AstraZeneca Nexium Prilosec PPI drugs settlement:
- Funds for operating costs: With post settlement funding, you can cover your firm’s day-to-day expenses such as rent, payroll, and other overhead costs, allowing you to focus on your clients’ cases without financial strain.
- No risk or credit check: Unlike traditional loans, post settlement funding does not require a credit check or personal guarantee. The funding is solely based on the case, and the settlement, which allows attorneys to secure financial assistance without putting their personal assets at risk.
How Does Post Settlement Funding Work?
The process of obtaining post settlement funding is simple and straightforward:
- Submit an application: Attorneys interested in post settlement funding can submit an application, providing details about their firm and the cases they are involved in.
- Case evaluation: A team of legal experts will review your application and evaluate the strength of the case.
- Funding offer: If approved, a funding offer will be made to your firm based on the evaluation of the case.
- Use funds as needed: Upon acceptance of the funding offer, attorneys can use the funds immediately for operating costs or however, they deem appropriate.
- Repayment only upon settlement: Once the case is successfully settled, repayment of the funding plus fees will be made from the settlement proceeds. If there is no successful outcome, there is no obligation to repay.

Post settlement funding is a valuable resource for attorneys involved in MDL’s and Class Action Lawsuits like the AstraZeneca Nexium Prilosec PPI drugs settlement. It can provide financial stability and flexibility during the long wait for a settlement, allowing attorneys to focus on their client’s cases without worrying about financial strain.
If you are an attorney involved in the Prilosec – Nexium settlement, consider exploring post settlement funding as an option to support your firm’s finances.
Post Settlement Funding is a Non-Recourse Transaction: We Accept All the Risk
Post settlement funding is a non-recourse transaction. This means you don’t need to worry about what might happen if the defendant suddenly can’t pay — we accept all risk of non-payment, meaning that you will still get to keep the money from your settlement advance if the defendant goes bankrupt or is unable to pay for whatever reason.
Fast, Hassle Free Application
In most cases, we can get your money in your hands in one week or less.
Our application is simple, straightforward, and easy to complete. Remember, this isn’t a loan, so there isn’t as much paperwork to go through. In most cases, we can approve your application and have your money deposited into your checking account in a matter of days.
If you are a lawyer involved in the Proton Pump Inhibitor lawsuits and would like to take out an advance on the contingency fees you are owed, Post Settlement Funding for Attorneys could be the right fit for you, please call one of our Post Settlement Funding specialists at 267-457-4540.
To apply online, simply CLICK HERE to fill out our quick application.
About The Authors
Balanced Bridge Funding offers legal funding solutions for plaintiff attorneys, attorneys, and law firms. To talk to one of our legal funding specialists about post settlement funding for attorneys, please call (267) 457-4540 or email info@balancedbridge.com.







